How to Keep your Brain Young with Antioxidants
How To Keep Your Brain Young with Antioxidants?

Being alive is a balancing act between making energy by combusting glucose with oxygen and generating ‘oxidant’ exhaust fumes that must then be neutralised. This process, known as oxidative stress, is a key contributor to ageing. Over time, if oxidants outpace your body’s ability to disarm them, damage accumulates in cells and tissues, including the brain.[8]
The brain is particularly vulnerable. It consumes a high proportion of the body’s oxygen, is rich in fats that are susceptible to oxidation, and has relatively limited antioxidant defences compared with other tissues. This makes maintaining an effective antioxidant defence system essential for long-term cognitive health.
However, this process is not fixed. You can influence it. Improving your intake of antioxidants and polyphenols, and supporting your body’s internal defence systems, can help shift the balance and support brain resilience over time.
To explore this idea further, watch the film ‘Keeping Your Brain Young with Antioxidants’ below.
The Science Behind Eating the Rainbow
You’ve heard it before, but the science behind it matters.
Different colours in plant foods reflect different polyphenols and antioxidant compounds, each with distinct biological effects. These compounds do more than just neutralise oxidants. Many also act as signalling molecules, influencing inflammation, blood flow, and cellular pathways linked to brain function and ageing.[9]
Mustard and turmeric, for example, are strong yellows. Dijon mustard is great, with no added sugar, and traditional English mustard is also beneficial. Turmeric, rich in curcumin, can be added to steam-fries, curries, or soups, grated into a tea, and even used in a turmeric latté.
Bright oranges such as butternut squash, sweet potato, and carrots provide carotenoids that support cellular protection. Tomatoes are particularly rich in lycopene, associated with reduced oxidative damage. Strawberries are a lower glycaemic fruit option, and peppers of all colours are rich in vitamin C, which plays a central role in antioxidant recycling.
Anything purple, magenta, or blue is especially valuable. From beetroot to blueberries, blackberries, and raspberries, these foods are rich in anthocyanidins, a class of polyphenols associated with improved vascular and cognitive function.
Strong greens remain foundational. Spinach, kale, Brussels sprouts, broccoli, watercress, rocket, asparagus, and green beans all contribute a wide spectrum of antioxidants, minerals, and phytonutrients that support detoxification and cellular defence.
Eating the rainbow is not a nice idea. It’s an essential part of upgrading and protecting your brain at any age.
The Best Fruits and Vegetables for Brain Health
But are there any particular vegetables or fruits that pack the biggest punch as far as polyphenols and antioxidants are concerned? Or, if you know you can’t or don’t like to eat a huge variety of fruit and veg, are there particular ones to focus on eating to get the most benefit?
Foods that are high in ‘polyphenols’, which refers to the structure of plant-based compounds, seem especially beneficial for protecting your brain. You might have heard of flavonoids in berries, quercetin in red onions, anthocyanidins in blue and red foods, and isoflavones in beans. These are all examples of polyphenols. Herbs and spices such as peppermint, basil, oregano, cumin, and curcumin in turmeric contain high levels of polyphenols and potent antioxidants.
But there are other criteria by which to judge a plant, including its ability to influence pathways linked to cellular ageing. Certain polyphenols, such as resveratrol, interact with sirtuin pathways involved in cellular repair and longevity.[10] For example, olives, blueberries, and kale support these processes.
Then there’s a group of compounds called ‘salvestrols’, generally only found in organic fruit, vegetables, herbs, and spices, which turn out to be anti-cancer.[1] They’re produced in plants as part of their self-defence system against invaders such as fungi. If the plant is sprayed with fungicides, it won’t produce them.
Taking all these factors into account, including the GL, antioxidants, polyphenols, salvestrols, and sirtuin-related activity, these are the dozen best-rated fruits and vegetables. (This list is not definitive. More and more research continues to reveal the healing power of nature’s fruits and vegetables.)
So, if eating the ‘rainbow’ feels like too much pressure or is difficult to achieve, think about incorporating some of the above into your meals each day.
Our Top 13 to Help Keep Your Brain Young
| Lowest GL | Antioxidant | Polyphenol | Salvestrol | Sirtuin Act. | |
| Olives | *** | *** | *** | *** | *** |
| Blueberries | *** | *** | ** | *** | *** |
| Kale | *** | ** | *** | *** | *** |
| Blackcurrants | ** | *** | ** | *** | *** |
| Broccoli | *** | ** | *** | *** | |
| Artichokes | *** | ** | *** | *** | |
| Cabbage (red) | *** | *** | ** | *** | |
| Asparagus | *** | ** | ** | *** | |
| Onions (red) | ** | * | *** | *** | |
| Avocado | *** | ** | ** | *** | |
| Apples | ** | ** | ** | ** | ** |
| Beetroot | * | * | *** | ||
| Cherries | ** | ** | ** |
Top Up Brain-Friendly Antioxidant Nutrients: Consider Supplementation
Antioxidants Work as a Network
Much like dementia prevention is a combination of the 8 domains, which all influence each other, antioxidants are part of a network keeping you healthy.
A number of key vitamins, as measured in food and in the blood, do correlate with decreased dementia risk. This is hardly surprising, since the brain is made of complex fats that can easily be damaged by oxidants. It makes sense that having a high intake of antioxidants would protect the brain from damage. Antioxidants disarm oxidants by teamwork. You need a combination of nutrients, not just vitamin C or vitamin E.
Evidence for Antioxidant Protection
A recent meta-analysis of all studies on factors that could prevent Alzheimer’s, by one of our Scientific Advisory Board members, Professor Jin Tai Yu of Fudan University in Shanghai, China, shows that ‘either a high vitamin E or C intake showed a trend of attenuating risk by about 26%’, making these nutrients ‘grade 1’ top level prevention risk factors.[2]
A study of 4,740 Cache County, Utah, older residents found that those supplementing both vitamin E and C cut their risk of developing Alzheimer’s by two thirds. A trend toward lower Alzheimer’s risk was also evident in those who took vitamin E supplements together with multivitamins containing vitamin C, but there was no evidence of a protective effect in those taking only vitamin E or vitamin C supplements alone, with multivitamins alone, or with vitamin B-complex supplements. Lowest risk was reported in those supplementing at least 1000 mg a day of vitamin C together with at least 1000 IU a day of vitamin E.[3]
However, vitamin E on its own doesn’t seem to work. In a double-blind study, people with mild cognitive impairment were randomly assigned to receive 2000 IU a day of vitamin E or placebo for three years. There were no significant differences in the rate of progression to AD between the vitamin E and placebo groups at any point.[4]
Building Your Antioxidant Defences
Your best bet is probably to both eat a broad spectrum of antioxidants and also supplement them. The older you are, the more you are likely to need. Key antioxidants are:
Vitamin A, C and E – associated with reducing Alzheimer’s risk
Lipoic acid – protects the memory-friendly neurotransmitter acetylcholine and dampens brain oxidation and inflammation.[5]
Glutathione[6] or N-acetyl cysteine[7] (NAC) – protects the brain and improves methylation, thus having potential in dementia prevention.
It doesn’t really make a lot of sense to give one without the others. All those listed above, vitamin C, E, glutathione, N-acetyl cysteine, Coenzyme Q10 and resveratrol, work together. There are many other team player ‘cousins’, from B vitamins to minerals such as magnesium, zinc and selenium.
The first step is to eat ‘whole’ foods, especially fresh plant foods that are more likely to contain these kinds of nutrients. However, there are some nutrients, such as vitamin C, for which just eating whole foods doesn’t guarantee you are achieving optimum nutrition.
Most nutritional therapists supplement extra vitamin C, and some supplement an all-round antioxidant supplement providing the nutrients listed above. There is very good logic, and supporting evidence, to do this, especially if you’re over 50 years old, even if there isn’t yet that definitive ‘randomised placebo controlled trial’.
Other key antioxidant nutrients include:
Vitamin A, C and E – associated with reduced Alzheimer’s risk
Lipoic acid – supports mitochondrial function and reduces oxidative stress and inflammation.[5]
Glutathione or N-acetyl cysteine (NAC) – supports detoxification, antioxidant recycling and methylation, with potential relevance for cognitive ageing.[6][7]
Coenzyme Q10 and resveratrol – support cellular energy and protection
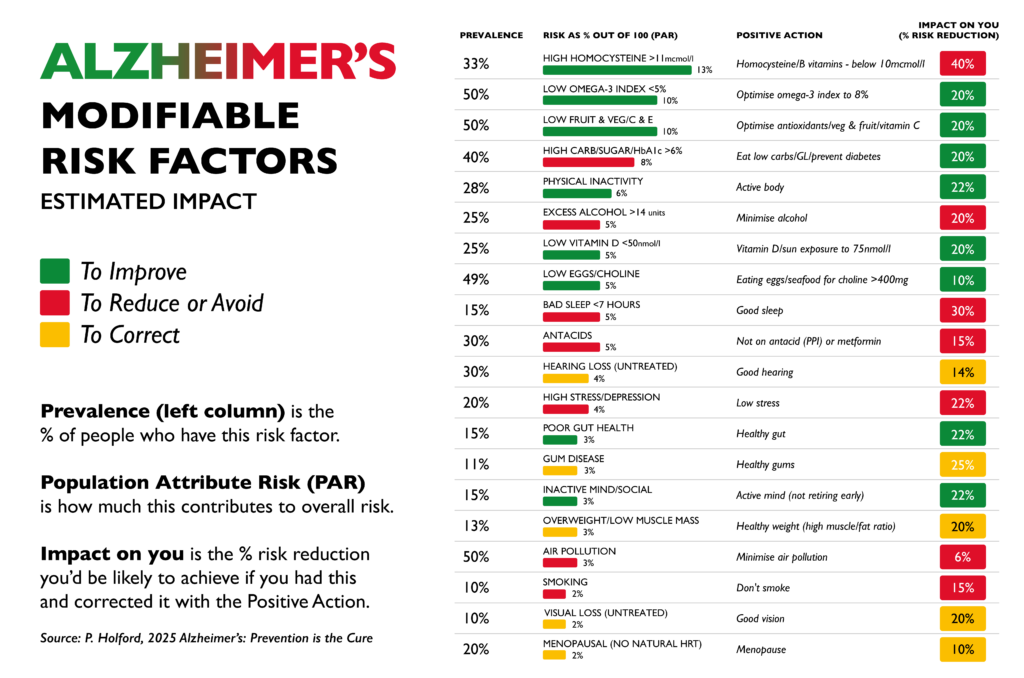
There are also important supporting nutrients, including B vitamins, which contribute to methylation and help regulate homocysteine, a compound associated with oxidative stress and cognitive decline when elevated.[12]
Why Food Comes First but Supplements May Still Be Needed
We are Food for the Brain, so the first step is always to eat whole foods, focusing especially on fresh plant foods that are naturally rich in antioxidants and polyphenols.
However, achieving optimal levels of certain nutrients through diet alone is not always guaranteed. Factors such as soil depletion, food storage, genetics, individual absorption, age, and increased physiological demand all play a role.
For this reason, targeted supplementation, particularly for nutrients such as vitamin C and those involved in the antioxidant network, may be beneficial. There is strong rationale, and supporting evidence, for this approach, especially in individuals over 50 or those with increased oxidative load. Read more about supplementation here.
From General Brain Health Advice to Personalised Insight
Why Antioxidant Needs Differ and Why Testing Matters
Eating a diet rich in colourful plant foods is a powerful place to start. But the real question is whether your unique body has the antioxidant capacity to meet your current level of demand.
Or, said another way, how do you know if you are eating enough to protect your brain and your future?
Oxidative stress is influenced by far more than diet alone. Age, stress, environmental toxins, blood sugar balance, genetic variations, nutrient status, and individual biochemistry all play a role. Two people can eat similarly and have very different levels of oxidative damage and antioxidant protection.
This is where testing becomes valuable.
One of the most informative markers is glutathione, often referred to as the body’s master antioxidant. It sits at the centre of your antioxidant defence system, helping to neutralise oxidative damage and recycle other antioxidants. If levels are low, it can indicate that your overall antioxidant capacity is under strain.
Rather than guessing whether you are getting enough antioxidant support, testing allows you to see what is actually happening inside your body. (Historically, glutathione has been hard to test, but we developed new technology with our lab partners to now be able to do this accurately with an at home finger prick blood test.)
Understanding Your Brain Health More Clearly
The DRIfT 5-in-1 test shows you what is actually happening inside your body, across the key drivers of cognitive decline, including oxidative stress, inflammation, blood sugar balance, nutrient status, and essential fats. It moves you beyond general advice and highlights exactly where your biggest risks and opportunities lie.
If your focus is antioxidant capacity, testing glutathione offers direct insight into whether your body is under oxidative strain and how well your defence system is functioning. Find out more here.
Alongside this, our free Cognitive Function Test provides a practical way to track how your brain is performing today, while contributing to ongoing research into what truly works in preventing cognitive decline.
Understanding your results allows you to move from general advice to a more targeted, personalised approach, so you can support your brain with greater precision and confidence.
When you join COGNITION and become a FRIEND, you get access to our new Digital education platform and our new COGNITION programme. You can also register FREE for both our monthly guest speaker webinars and our monthly live COGNITION Coaching – this is an hour live with our Food for the Brain health coaches and there is a Q&A at the end (you also get access to the past coaching workshop recordings).
On April 22nd April, our COGNITION Coaching Workshop is all about antioxidants and attendees will get a mini-ebook on antioxidants and 2 delicious, anti-oxidant rich recipes. You can become a FRIEND by clicking the link below:
References:
[1] Potter, G.A. & Burke, M.D. (2006) Salvestrols – Natural Products with Tumour Selective Activity. Journal of Orthomolecular Medicine. 21(1): 34-36.
[2] Yu JT, Xu W, Tan CC, et al. Evidence-based prevention of Alzheimer’s disease: systematic review and meta-analysis. J Neurol Neurosurg Psychiatry. 2020;91(11):1201–1209.
[3] Zandi PP, et al. Reduced risk of Alzheimer disease in users of antioxidant vitamin supplements. Arch Neurol. 2004;61:82–98.
[4] Petersen RC, et al. Vitamin E and donepezil for the treatment of mild cognitive impairment. N Engl J Med. 2005;352:2379–2388.
[5] Maczurek A, et al. Lipoic acid as an anti-inflammatory and neuroprotective treatment. Adv Drug Deliv Rev. 2008;60(13-14):1463–1470.
[6] Pocernich CB, Butterfield DA. Elevation of glutathione as a therapeutic strategy. Biochim Biophys Acta. 2012;1822(5):625–630.
[7] Hara Y, et al. Evaluation of N-acetylcysteine for cognitive ageing. J Prev Alzheimers Dis. 2017;4(3):201–206.
[8] Halliwell B. Oxidative stress and neurodegeneration. J Neurochem. 2006;97(6):1634–1658.
[9] Spencer JPE. The impact of flavonoids on memory. Chem Soc Rev. 2009;38(4):1152–1161.
[10] Baur JA, Sinclair DA. Therapeutic potential of resveratrol. Nat Rev Drug Discov. 2006;5(6):493–506.
[11] Sekhar RV, et al. Glutathione synthesis is diminished in older adults. Am J Clin Nutr. 2011;94(3):847–853.
[12] Smith AD, Refsum H. Homocysteine, B vitamins, and cognitive impairment. Annu Rev Nutr. 2016;36:211–239.