Dr Tommy Wood: Why Brain Health Needs a Systems-Based Approach
Dr Tommy Wood: Why Brain Health Needs a Systems-Based Approach

Dr Tommy Wood brain health research shows that we now know more than ever about protecting the brain, yet many people still feel overwhelmed by disconnected advice.
A headline about blood sugar here. A podcast on omega-3 there. A post about sleep, a study on exercise, a warning about stress.
Useful, yes. But also fragmented.
What Dr Tommy Wood brain health research offers is something more valuable than another isolated tip. He brings a framework.
As Head of Research at Food for the Brain, Associate Professor of Pediatrics and Neuroscience at the University of Washington, and author of the forthcoming book The Stimulated Mind, Tommy brings something rare to this field. He helps connect the dots. His work focuses on how brain health is shaped across the lifespan and why protecting cognitive function means thinking in systems, not silos.
Dr Tommy Wood Brain Health Framework
One of the reasons brain health can feel confusing is that it is often presented as a list:
A long list of risk factors. Nutrients to take, habits to adopt, or a long list of things to avoid.
But the brain does not work like a checklist: it works as a living, dynamic system. It responds to what it is supplied with, how it is used, and whether it has the conditions it needs to recover and adapt.
That is the core idea behind a systems-based approach to brain health.
Instead of asking “What is the one thing that protects the brain?”, it asks three bigger questions:
- Does the brain receive enough oxygen, nutrients and stable energy to function well?
- How well is it being stimulated to stay active, adaptable and resilient?
- Are the foundations for repair and recovery in place, such as good sleep, stress regulation and metabolic health?
When you look at brain health this way, something important changes.
Many people understandably hope for a “magic bullet”. One supplement. One diet. One breakthrough drug that protects the brain.
But science tells a different (more hopeful) story. Cognitive resilience is shaped by the interaction between many systems in the body. Blood flow, nutrient status, sleep, movement, learning, stress and social connection all influence how the brain functions and adapts.
The encouraging part is that this gives us many entry points for change. You do not have to get everything perfect, and small improvements across several areas can work together to create meaningful protection for the brain over time.
That is why a systems-based approach is not more complicated. In practice, it is far more empowering.
Why this matters for prevention?
One of the most important insights in Tommy’s work is that cognitive decline does not happen in isolation.
The brain changes in response to how we live. Blood flow, nutrient status, movement, learning, sleep, stress and social connection all shape how the brain ages and how resilient it remains.
This is also why Tommy’s forthcoming book, The Stimulated Mind, focuses on the interaction between stimulation, sleep and nutrient supply as key drivers of lifelong brain health.
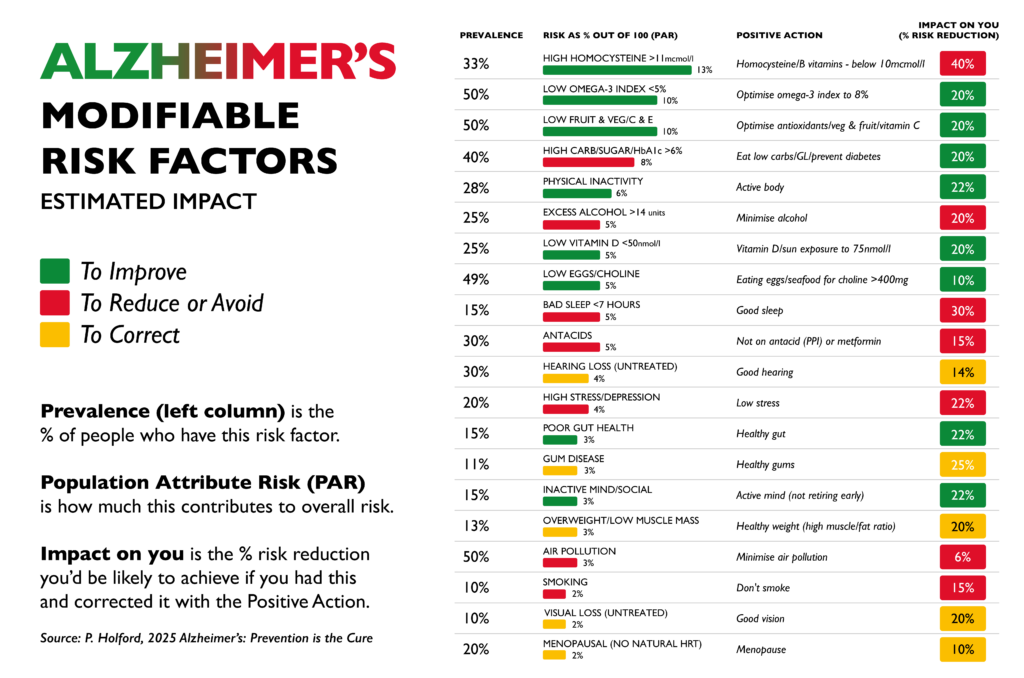
This perspective is closely aligned with the mission of Food for the Brain. For years we have been communicating that dementia risk is not fixed. By understanding and addressing modifiable factors across the lifespan, it is possible to protect your mind and keep the brain healthier for longer.
Tommy’s leadership as Head of Research helps bring greater scientific clarity to this systems-based approach to prevention.
Watch: A systems-based approach to cognitive function
If you want to hear Tommy explain this approach in more depth, his lecture from the Upgrade Your Brain Conference is one of the clearest introductions we have shared.
Watch the lecture below.
What Tommy offers is not another list of things to do. It is a framework. A way to understand why so many different factors matter and how they fit together.
Learn More About Dr Tommy Wood Brain Health Research
If this approach resonates with you, Tommy will be going deeper in our upcoming webinar, How to Keep Your Brain Young.
In this session he will explore the science of cognitive resilience and share practical ways to stimulate the brain, support long-term brain health and reduce lifetime dementia risk.
Food for the Brain will host the webinar on 19th March 2026 at 6 pm GMT.
Tommy’s new book is available to pre-order

Tommy’s new book, The Stimulated Mind, is also available to pre-order now.
In it, he explores how the brain develops, adapts, and stays resilient across the lifespan, and why everyday factors such as movement, learning, sleep, nutrition, and social connection play such an important role in shaping long-term brain health.
Pre-order your copy today before the book releases on 26th March 2026.
Final thought
For many people, the challenge with brain health is not a lack of information. It is knowing where to start.
When advice comes as scattered tips, nutrients, tests, and lifestyle changes, it can feel difficult to turn good science into clear action.
What Tommy’s work offers is a framework: a way of understanding how the different pieces of brain health fit together and why small changes across several areas can have a powerful cumulative effect.
At Food for the Brain, this systems-based approach sits at the heart of COGNITION, our brain upgrade programme.
COGNITION helps you understand your personal risk factors, learn the most important steps to protect your brain, and put them into practice with guidance and support.
The programme has recently been upgraded and is now available globally for just £5 per month or £50 a year, making it accessible to anyone who wants to take their brain health seriously.
If Tommy’s lecture resonates with you, COGNITION is the natural next step.
Because protecting your brain is not about doing everything perfectly.
It is about understanding the system and taking the next right step.