Alzheimer’s: Why Prevention is Better than Cure
Everybody wants a cure for Alzheimer’s.
The medical industry has spent around $100 billion searching for one and so far, come up relatively empty-handed with over thirty failed drug trials. The focus has been on drugs that lower two of the chemical compounds associated with Alzheimer’s and dementia in general – amyloid and p-tau, a pair of messed up proteins that can lead to plaques in the brain and tangled nerves. There is a third compound – an amino acid called homocysteine – that becomes toxic if you have too much, that the drug industry and the Alzheimer charities don’t talk about, for reasons that will become clear.
The actual clinical measures that are used to diagnose Alzheimer’s are a decline in cognitive function and shrinkage of the central area of the brain called the medial temporal lobe. Both changes in cognitive function and brain shrinkage can be picked up thirty years before a diagnosis of Alzheimer’s is made.
So now a £10 million study is underway to see if a blood test for p-tau, or amyloid, will ‘predict’ if you are more likely to develop the disease and there are plans for a major programme to identify those at risk so they can be treated as early as possible. This sounds sensible but there are serious drawbacks. To begin with not everyone with raised p-tau or amyloid go on to develop Alzheimer’s.
This means, as a recent article in the New York Times entitled, ‘Apparently healthy but diagnosed with Alzheimer’s,’ pointed out, people without a diagnosis or no brain scan showing shrinkage, could well be offered new drug treatments that are so far, only marginally better than placebos but can have awful adverse effects.
These include brain bleeding or swelling which has occurred in more than one in four in the last two drug trials and resulted in seven deaths. Medical agencies in the US, EU and UK are reluctant to licence their use but are under a lot of pressure to do so.
So thousands of desperate people with early stage Alzheimer’s or cognitive decline, hoping for a cure, are queuing up to join these drug trials because they perceive these drugs, that so far come with little or no benefit plus highly unpalatable side effects, are a better alternative than doing nothing.
—
There are alternatives – you just haven’t been told about them!
But are there really no alternatives? Well, none that patients are routinely told about. They involve changes in diet and lifestyle that are very likely to improve your overall health, including that of your brain, and very unlikely to cause damaging side effects.
Almost all money for research, pledged by governments and raised by Alzheimer’s charities, is going in the direction of drug treatments. Alzheimer’s Research UK’s (ARUK) website says “we exist for a cure”. Most of the money is going toward amyloid and p-tau related research, neither of which have been established as causal. In other words, high levels may just be a consequence of the disease process.
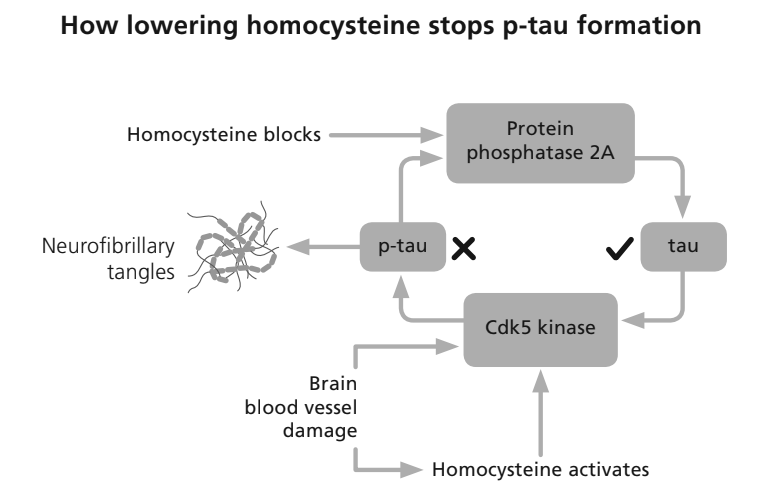
The same is not true for raised blood levels of homocysteine. If levels rise in the brain, it shrinks faster and cognitive abilities decline. If it goes down, they improve, and brain shrinkage slows. This means that it is causing the damage and so would logically be a target for treatment. The only way to do it, however, is with high dose B vitamins (B6, B12 and folate). Several gold standard, placebo-controlled trials have found this to be very safe and effective. But this approach is not patentable and so yields nothing like a drug profit.
—
This is a much better biomarker than p-tau
But the benefits of treating homocysteine don’t stop there. It is a much better biomarker of risk for Alzheimer’s than plaque and p-tau both because it is more easily measured and more safely lowered. And when it is lowered, unlike those two, it actually improves cognitive function and slows brain shrinkage by as much as two thirds. It also helps to stop p-tau formation.
Routinely checking homocysteine levels could prevent thousands of cases. Just doing this “could save costs to the UK economy of approximately £60 million per year,” says Dr Apostolos Tsiachristas, Associate Professor in Health Economics at the University of Oxford. His research also estimated it would promote healthy longevity, adding 14 years to life expectancy.
About half of people over 65 have a homocysteine level above 11 mcmol/l, which is where it starts to become damaging. In one study a third of those treated ended the study with no clinical dementia rating, meaning they could no longer be diagnosed with cognitive impairment. Those with sufficient omega-3 DHA, which is the most important structural fat in the brain, had 73% less brain shrinkage compared to placebo when given the B vitamin treatment. In contrast, in the last anti-amyloid treatment trial, brain shrinkage accelerated by about a fifth in those getting the drug, compared to placebo and not one person achieved a clinical dementia rating of zero.
It should be clear by now, after decades of scientific research that amyloid plaque is not a cause of Alzheimer’s, but a consequence. The same is likely to be true for p-tau tangles.
As an analogy, consider your teeth. Is plaque the cause of tooth decay? Sure, flossing your teeth and getting the plaque scraped off by the dental hygienist helps, but what causes the plaque? The answer is a bad diet – in this case, one high in sugar and low in fibre. Despite fifty years of research, there is no ‘cure’ for tooth decay, but it can be prevented. The same concept applies to Alzheimer’s, which is as preventable as tooth decay with the right diet and nutrition and lifestyle – which also happens to include less sugar and more fibre.
—
Alzheimer’s Prevention
How preventable is Alzheimer’s? It accounts for two thirds of dementia cases. The most conservative figure is 40% . More optimistic estimates say around 80%. Since only one in a hundred cases is caused by genes Alzheimer’s may be entirely preventable in those 99% who do not have the rare causative genes and act early enough to optimise all diet and lifestyle factors. It is not an inevitable consequence of the ageing process as evidenced by the fact that the majority of people don’t get it.
Why the difference in figures? It’s all to do with what is or isn’t included in prevention studies. The most widely used review for dementia prevention in the UK is the 2020 report of the Lancet Commission, authored by Professor Gill Livingston. Both this and the first edition in 2017 failed to even mention homocysteine, despite being repeatedly sent all the evidence of the undeniable beneficial effects of homocysteine-lowering B vitamins by the Oxford Project to Investigate Memory and Ageing (OPTIMA) at the University, headed by former Deputy Head of Medical Science, Professor David Smith.
This is a major and damaging error and has led to the widespread belief that B vitamin supplements are not part of the usual list of preventive actions. But it should be corrected, especially considering that a US National Institutes of Health study attributes 22% of the risk of Alzheimer’s to raised homocysteine. Also, the best study of all, looking at 396 studies in total, published in 2020, concluded: ‘Homocysteine-lowering treatment seems the most promising intervention for Alzheimer’s disease prevention.’
Other prevention studies you may have read are possibly based on data from the UK Biobank. This major research data bank also ignores homocysteine, not for any malevolent reason but simply because it wasn’t measured when it was enrolling people. So, one of the single biggest risk factors and arguably the simplest to change, is repeatedly ignored.

Given that a conservative half of Alzheimer’s cases could be prevented, shouldn’t half the available research money be spent on prevention?
This certainly doesn’t happen at the moment. Of the three leading charities, two spend nothing on prevention. ARUK claims to spend 5% but none of this goes towards B vitamins or other brain-friendly nutrients such as omega-3 or vitamin D. They, too, ignore homocysteine, and the beneficial effects of lowering it with B vitamins, as first shown in a 2010 Oxford University study they actually helped fund!
—
1+1=3
Prevention studies are almost always going to underestimate (never overestimate) the power of prevention due to excluding risk factors, but also because they largely ignore the ‘1+1=3’ compounding impact of interactive risk factors. B vitamins, for example, don’t work without sufficient omega-3 and omega-3 fish oils don’t work in people with raised homocysteine, because of a lack of B vitamins. This has been shown in four trials – in the UK, Holland, Sweden and China. The combination of B vitamins given to people sufficient in omega-3 DHA improved the reduction in brain shrinkage from 53% to 73%.
Pollution exposure is a risk factor but, in those with lower homocysteine, this effect is much reduced. Poor sleep is a risk factor, but less so in those who exercise.
For the past five years leading UK researchers, led by neurologist Professor Peter Garrard, who is the Director of the dementia research group in the St George’s, University of London Neuroscience Research Section, have tried to get funding to test the most promising combination – B vitamins and omega-3 – to no avail. Such a trial is badly needed and would cost a fraction of that being spent on amyloid or p-tau.
So. What if a person does everything right – enough B vitamins to keep homocysteine low, sufficient omega-3, low sugar, high fibre diet, enough vitamin D (Alzheimer’s is four times less likely in those with sufficient vitamin D), and an active physical, intellectual and social lifestyle, plus good sleep and not too much stress?
The only ongoing study and database that assesses all these risk factors as well as including blood tests of four critical biomarkers, homocysteine, omega-3 index, vitamin D and HbA1c, which measures glucose control, is our COGNITION Biobank. We describe it as ‘citizen science’ because anyone can get involved doing a free online Cognitive Function Test, filling in a questionnaire about their diet, lifestyle and medical history, and sending in a blood sample from our home test kit available for purchase.

So far, over 400,000 people have done our test. But, unlike the £10 million trial, funded by the People’s Lottery, the Gates Foundation, ARUK and the Alzheimer’s Society, it gets no funding. It is literally funded by our citizen scientists who chip in £50 a year and pay for their own tests. Their message is simple: prevention is better than cure – don’t jump.
Action:
- Take the FREE Cognitive Function Test here
- Order your at-home blood tests here
- Support our research and become a FRIEND here
- Participate in Alzheimer’s Prevention Day
—
A world-class group of Alzheimer’s prevention experts are launching Alzheimer’s Prevention Day on May 15th. See alzheimersprevention.info.
(The 3-minute Alzheimer’s Prevention Check goes live on May 1st).
—
Thank you for reading!
Food for the Brain is a non-for-profit educational and research charity that offers a free Cognitive Function Test and assesses your Dementia Risk Index to be able to advise you on how to dementia-proof your diet and lifestyle.
By completing the Cognitive Function Test you are joining our grassroots research initiative to find out what really works for preventing cognitive decline. We share our ongoing research results with you to help you make brain-friendly choices.
Please support our research by becoming a Friend of Food for the Brain.
References & Info
These are key papers regarding stated facts in this article.
New York Times article: https://www.nytimes.com/2024/03/04/health/alzheimers-amyloid-diagnosis.html
Homocysteine and p-tau: https://foodforthebrain.org/the-p-tau-delusion/
Donanemab review in the British medical Journal: BMJ 2023;382:p1852
Telegraph reports 7 deaths and brain shrinkage: https://www.telegraph.co.uk/news/2024/02/19/alzheimers-drugs-shrink-brain-scientists-warn/
Health economics of B vitamins: Tsiachristas A, Smith AD. B-vitamins are potentially a cost-effective population health strategy to tackle dementia: Too good to be true? Alzheimers Dement (N Y). 2016 Aug 11;2(3):156-161. doi: 10.1016/j.trci.2016.07.002. PMID: 29067302; PMCID: PMC5651357.
Omega-3 and B vitamin interactions and studies: Smith AD, Jernerén F, Refsum H. ω-3 fatty acids and their interactions. Am J Clin Nutr. 2021 Apr 6;113(4):775-778. doi: 10.1093/ajcn/nqab013. PMID: 33711096.
Less brain shrinkage and cognitive decline with B vitamins and sufficient omega-3: Jernerén F, Elshorbagy AK, Oulhaj A, Smith SM, Refsum H, Smith AD. Brain atrophy in cognitively impaired elderly: the importance of long-chain ω-3 fatty acids and B vitamin status in a randomized controlled trial. Am J Clin Nutr. 2015 Jul;102(1):215-21. doi: 10.3945/ajcn.114.103283. Epub 2015 Apr 15. PMID: 25877495; see also Oulhaj A, Jernerén F, Refsum H, Smith AD, de Jager CA. Omega-3 Fatty Acid Status Enhances the Prevention of Cognitive Decline by B Vitamins in Mild Cognitive Impairment. J Alzheimers Dis. 2016;50(2):547-57. doi: 10.3233/JAD-150777. PMID: 26757190; PMCID: PMC4927899.
NIH Alzheimer’s prevention review: Beydoun MA, Beydoun HA, Gamaldo AA, Teel A, Zonderman AB, Wang Y. Epidemiologic studies of modifiable factors associated with cognition and dementia: systematic review and meta-analysis. BMC Public Health. 2014 Jun 24;14:643. doi: 10.1186/1471-2458-14-643. PMID: 24962204; PMCID: PMC4099157.
Meta-analysis of 396 studies favouring homocysteine-lowering B vitamin treatment: Prof Yu study Yu JT, Xu W, Tan CC, Andrieu S, Suckling J, Evangelou E, Pan A, Zhang C, Jia J, Feng L, Kua EH, Wang YJ, Wang HF, Tan MS, Li JQ, Hou XH, Wan Y, Tan L, Mok V, Tan L, Dong Q, Touchon J, Gauthier S, Aisen PS, Vellas B. Evidence-based prevention of Alzheimer’s disease: systematic review and meta-analysis of 243 observational prospective studies and 153 randomised controlled trials. J Neurol Neurosurg Psychiatry. 2020 Nov;91(11):1201-1209. doi: 10.1136/jnnp-2019-321913. Epub 2020 Jul 20. PMID: 32690803; PMCID: PMC7569385.