If you ask the man on the street what’s driving Alzheimer’s, they’ll probably say it’s in the genes or that it is just what happens when you age.
Now, neither of these statements are strictly true.
Alzheimer’s is a largely preventable disease.
The big question though is WHAT exactly needs to be done and HOW do we do this?
Dr. Tommy Wood is an Assistant Professor of Pediatrics and Neuroscience at the University of Washington in Seattle. His research program focuses on factors associated with brain health and function across the lifespan. He received his undergraduate degree in Natural Sciences from Cambridge in 2007, a Medical Degree from Oxford in 2011, and a PhD from the University of Oslo in 2017. Alongside his academic training, Tommy has provided Performance Consulting for Olympians and world champions in a dozen different sports. He is a founding trustee of the British Society of Lifestyle Medicine and associate editor for the journal Lifestyle Medicine.
Dr Kristina Curtis is an Expert in Digital Behaviour Change Interventions (DBCIs). She has a multidisciplinary background spanning across industry, research, teaching, training and consultancy. Her primary research interests are in the development and evaluation of mHealth (mobile health) interventions, in particular how the convergence of behavioural science and UX design promotes effective engagement.
Interview:
Diagnosis & the difference between Alzheimer’s, dementia and mixed dementia.
My guests today are tackling the two fundamental questions.
- Firstly, what are the positive and negative behaviours, diet lifestyle and environment that both either drive dementia or prevent it?
- And then the big question is, how do you encourage people to change those behaviours?
So we have a system Professor, Tommy Wood from the University of Washington about the ‘what’. Then to Dr. Kristina Curtis, a behavioural scientist and honorary lecturer at the University College London who heads the applied behaviour change team about how to affect behaviour change.
Now, your background in relation to the brain is broad I see, from your time at Cambridge and Oxford University then Norway and now as assistant professor of Paediatrics and Neuroscience at the University of Washington. I believe you’ve also brain-trained Formula One drivers. Tell us about your background and how you became involved in this challenge to prevent Alzheimer’s.
I essentially fell into neuroscience initially, around 20 years ago when I was an undergraduate at Cambridge. I spent a summer in the neonates neuroscience lab looking at brain injury in babies. This is still partly what I do 20 years later.
In between, I then went to medical school where I trained as a doctor and I also developed a lot of interest in other neurodegenerative conditions. For instance, I spent a lot of time looking at a systems approach to multiple sclerosis because my step brother was diagnosed with multiple sclerosis when he was a similar age to me in his mid 20s.
Then, as I worked through my PhD in my early formal academic career as a professor, I worked increasingly with athletes as a performance consultant. So anything related to their cognitive and physical performance which could be sleep, diet and other stresses that they are frequently exposed to. So, as this story of my career comes together, I work with brain injury at the beginning of life, then as I work with athletes, I do more around concussions and traumatic brain injury. And then you think, well, there were all these factors that start early in life, probably even before you’re born, that create this trajectory of how your brain functions. And that continues as you get older. So then, that makes me think about, how do we intervene at any life stage to ensure that we have cognitive function that lasts as long as possible, ideally preventing significant cognitive decline and dementia in old age. Then you start to see that these same risk factors are important at every stage so that you can start to build this idea of what the brain needs, and then you can figure out how you might want to intervene.
I was just interviewing Professor Michael Crawford, age 93, and using some sort of quantum physics, he’s worked out how the photons that hit our eye turn into the image that we actually see. And I remember at the same age of 93, filming Linus Pauling on his theory of lipoprotein A. So I’ve witnessed people well into their 90s as sharp as a razor.
But before that, a little bit of background, what is the difference between Alzheimer’s and dementia overall, and we also hear about vascular dementia?
Dementia is essentially a catch-all diagnosis for when an individual has reached a point of cognitive decline where they are no longer able to perform regular daily tasks or usually look after themselves. So it’s a clinical diagnosis based on overall cognitive functioning.
Then you might ask, Well, what’s causing this dementia in an individual and then we we have the sub categories, so Alzheimer’s dementia, which, if you asked Alzheimer himself, apparently he was unsure whether they should all be classified as the same thing, but that’s a sort of semantic argument.
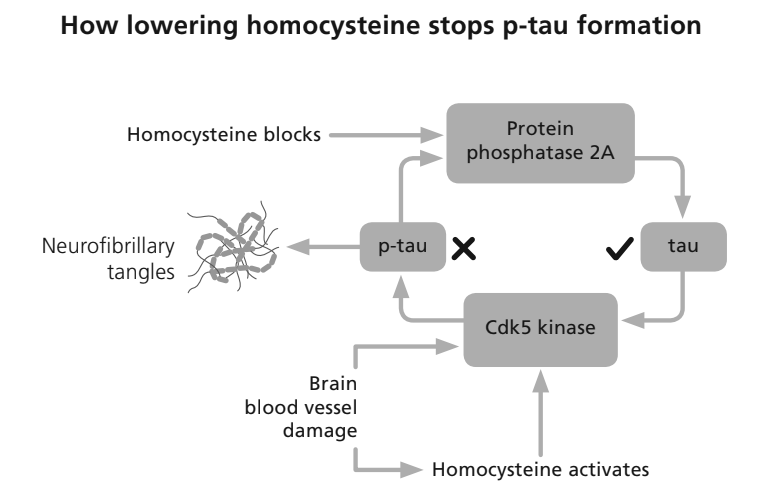
But what we would call Alzheimer’s clinically makes up maybe 60 to 80% of dementia. Vascular dementia, which is more directly focused on blood supply to the brain, makes up something like 5 to 10%. Then there are other dementias that have very special effects on very specific parts of the brain but overall, their effect on broad cognitive function eventually ends up in a similar place.
So with Alzheimer’s, dementia is often thought to be this continuous, gradual decline in function. Whereas with vascular dementia, what is often thought to happen is you have these very small strokes that happen throughout the brain. And each time that happens, you lose a portion of function, so you have more of this step change over time. However, in reality, there tends to be lots of overlap between say vascular and Alzheimer’s dementia, because blood supply to the brain is also important, for Alzheimer’s and sometimes it can be difficult to differentiate between the two.
And how is Alzheimer’s diagnosed as you know, in that form of dementia?
Usually, it’s going to involve some cognitive function tests. So there are lots of standardised tests like the MACA, MMSE appeal – you may have heard of these. But there’s a whole range of tests that you’re usually going to do in person with a neurologist or an old age psychiatrist and part of it is going to be a diagnosis of exclusion. So you want to make sure that somebody doesn’t have something else going on, like a very significant vitamin deficiency or other things that can be significant like depression as they can look like significant cognitive decline or dementia, but they aren’t necessarily the same thing. So part of it is going to be excluding these other causes. Then you will probably do some brain scans and look at how the brain looks either on a CT scan or an MRI and then that, in conjunction with cognitive function, and some part of the medical history is going to tell you this is likely Alzheimer’s dementia. Often, we hear of someone being diagnosed with mixed dementia.
Is that mainly part Alzheimers, part vascular?
Yes, that’s the most common combination, but if you think about Lewy body dementia, or Frontotemporal dementia, these can also occur in different combinations in different people. But usually, because of the overlap in terms of the risk factors, Alzheimer’s with some vascular component is relatively common.
Preventing Alzheimer’s
Now, if a person could change all their circumstances, and we’re sort of talking from early life as well, – their diet, their lifestyle, the environment that they’re born into, and their education, to what extent could we say that Alzheimer’s is a preventable disease?
It depends on how you want to try and tackle the question. But if you can change everything about an individual’s circumstances other than their genetics, then the vast majority is preventable. Depending on who you ask, some will say that it’s maybe 40 to 50%. Others will say that it’s up to maybe 70 to 80%. In some populations around the world, usually hunter-gatherer or indigenous populations, dementia is almost entirely unheard of. So it’s definitely possible that there are some combinations of environment and genetics where dementia just doesn’t occur and in that scenario, it is entirely preventable.
Now, do these same prevention steps, which obviously we’re going to go into, also prevent vascular dementia? And if so, if we put Alzheimer’s and vascular and a chunk of mixed dementia together, are we talking about 80% of dementia for example, being potentially 80% preventable?
Yes. So, as I mentioned briefly earlier, there’s a lot of overlap between the risk factors for vascular dementia. And those are also related to cardiovascular disease risk factors. So things that affect your body’s ability to move blood to the places that you want it to move to, the health of your blood vessels. There is a lot of overlap between those risk factors or things that affect negatively and those are risk factors for Alzheimer’s disease.
In fact, cardiovascular disease and poor vascular health are risk factors for Alzheimer’s dementia more directly. So if we think about the upper end of the subcategories of dementia that, 80% is Alzheimer’s disease, 10% is vascular dementia, which based on estimates could be up to 90% then yes, up to 90% of dementia is probably at least 70 ish to 80 ish percent preventable.
How important a role do genes play?
It’s often said that early onset Alzheimer’s, which is defined as before the age of 65, is that small genetic part, but a study that came out earlier this year questioned that. It was on the basis of data from the UK Biobank and it actually said that many of the same factors that are driving dementia or Alzheimer’s later in life are also present in those who develop a diagnosis before 65. So that sort of de-emphasises the genes to some extent.
Is this true, that early onset is much the same phenomena that we’re looking at.
So traditionally, early onset Alzheimer’s, is thought to be almost entirely genetic, and driven by single significant, high penetrance genetic mutations that result in early cognitive decline. In Alzheimer’s dementia, which is thought to be more similar to what Alzheimer himself was describing when he first described the disease, it was thought to be maybe five to 10% of dementia. Now, as Alzheimer’s diseases become more prevalent, it’s maybe around 1% of cases. However, I certainly know clinicians who have patients who have one of these mutations in a presenilin gene, or the amyloid precursor protein gene and by attending to lifestyle factors, they managed to maintain good cognitive function late into life. So even if you have some of these mutations, which are thought to almost definitely lead to Alzheimer’s disease, there does seem to be an interaction with lifestyle and other health related factors.
Then, at the same time, we’re seeing earlier diagnosis of non-monogenic Alzheimer’s disease, which is what this paper you described, was talking about. And that’s probably because the health of the population is declining, such that we’re seeing pre-diabetes earlier, we’re seeing people that are less physically active, maybe they’re less cognitively stimulated. All these other risk factors are adding up and they’re affecting our brains earlier in life. So now there’s more of a mix, in early onset Alzheimer’s disease, as those risk factors become prevalent earlier in life.
Diagnosed with Alzihemrs at age 19? A growing tsunami of cognitive decline
I saw a report from China, of a man aged 19 diagnosed with Alzheimer’s, it said it was non-genetic, so presumably, they tested and there wasn’t the Apoe gene or the pre Senlin gene, I can’t confirm that. But can it really happen that fast, this cognitive decline that we are seeing that is so prevalent?
If you think about cognitive function as a trajectory, over time, there are three or four different components to that.
So first of all, as you get older, cognitive function generally peaks on average, towards the end of your formal period of education. So if that education extends into university or maybe a graduate degree, then that’s going to peak sometime in your 20s or early 30s. But if you don’t even complete secondary school or high school, then it’s going to peak much earlier, and it’s going to have a much lower peak. After the peak, then you have a period of decline, and the speed of that decline depends on a number of factors, the majority of them related to ongoing stimulus to the brain as well as a whole host of health-related and nutritional factors, as you know very well. So there’s certainly a possibility where if you had a low peak of cognitive function, and that may even be based on something like maternal nutrition and epigenetics related to the health of your parents, so you had a low peak, and you have poor overall health, you’d have a more rapid decline. It’s certainly possible that this could happen very early in life, though, thankfully, at least right now, that’s very rare.
Yeah, we’ve seen the youngest with type two adult onset diabetes age three, so quite extraordinary.
So is Alzheimer’s, just the tip of the iceberg of a growing tsunami of cognitive decline that is likely to start happening earlier?
I say no, because having had lots of conversations, including with one of your close colleagues and mentors, David Smith. When you look at the specific prevalence of Alzheimer’s disease, it has not increased over time. And in fact, in some populations, it’s decreased. It’s probably decreased a little bit more in men, because we focus very heavily on cardiovascular disease risk factors, which are more important in men in terms of causes of death.
And that has come with some improvement in terms of age-specific incidence of Alzheimer’s disease, when the reason why Alzheimer’s disease is becoming more prevalent, and it’s going to double or triple in the next few decades, is because we’re living longer. So there’s a combination of we’re living longer, and in general, our health is declining.
So you could say, there’s this oncoming wave of Alzheimer’s disease, and that is expected. But we know that if we target specific risk factors, that doesn’t have to be the case. So I don’t think it’s all bad news.
Our Citizen Science Research Team
Now, you’re the principal investigator at Food for the Brain heading, the research and the science there. So what does that involve?
What’s your goal as a principal investigator?
One of the most important tools that we have when we’re looking at diseases or specific functions or health of the population are our population datasets, and there are lots of these.
Governments around the world organise them, there’s Haynes in the US, there’s the UK Biobank in the UK that people have heard of, and you collect lots of data from lots of people, and at that kind of scale, you can start to maybe pick up relationships that you couldn’t otherwise.
When we think about the risk factors for cognitive decline, and dementia, specifically, none of the datasets that exist so far are really designed to focus on that. So one of the most important ideas is to collect data, specifically related to risk factors for Alzheimer’s, dementia, and hopefully those that are modifiable. So it’s kind of a citizen science idea, right? We’re going to people who are interested in this or maybe interested in this for their loved ones. They are providing their data and part of it is to help understand their own risk, but then allowing us to look at a wide variety of risk factors.
There are over a hundred questions in a questionnaire that look at different risk factors and we have the cognitive function test and blood tests. So then, when you have very large datasets like this, we can start to look for complex interactions between risk factors, and we can figure out which risk factors are most important. These are things that still need to be done as it pertains to most Alzheimer’s disease. I think, hopefully, Food for the Brain is going to be in a good position to help people understand that.
Figuring out the perfect food and lifestyle combo – what matters more!
So what’s your ultimate goal from this data set – to find the sort of perfect diet lifestyle combo?
I think that there are two main things. In reality, I think that we know what is going to be best for most people. If we look across all the evidence, we probably know that already we can have a good guess of what you should aim for.
When you say, here are the 50 things you need to fix to improve your brain health. There’s just an overwhelming amount of information – you don’t know where to start, what should I do?
First, what’s most important to me, what’s most impactful? So figuring out which factors an individual should focus on first, which gives the biggest bang for their buck is what’s going to be a big, big part of this. Then the million dollar question is how do you actually change a person’s often highly entrenched behaviour, especially if we want to reach hundreds of 1000s across a digital platform?
So Dr. Kristina Curtis, welcome.
You’re a behaviour change expert and an associate of UCLA Centre of Behaviour Change and founder of Applied Behaviour Change, which focuses on digital health programmes to help prevent and manage chronic conditions.
Can you explain a bit about your background and the reason you are interested and involved in this Alzheimer’s prevention project?
After my psychology degree, I had a few years out working in industry because I hadn’t found the area of psychology that I wanted to specialise in. And so this led me to return to studies to specialise in health psychology, which is all about health behaviours. And then immediately afterwards, I then started a PhD, which really culminated my experience back then, which was web to technology, with my academic knowledge of health psychology at the Institute of Digital Healthcare at Warwick University.
My PhD was really focused on how we would embed behaviour change models into mobile health apps and digital health interventions. And really, I feel passionate about preventing chronic conditions and particularly Alzheimer’s disease, as there are a lot of misconceptions about it. And we’ve talked a lot about them today. And in terms of many people think it’s a symptom of ageing, and largely down to our genes. Whereas, as we’ve heard, we can do a lot to reduce our risk by adopting healthier habits.
How do we implement these ‘risk-reducing’ behaviours and habits?
And what are you doing in the cognition project at Food for the Brain? What’s your game plan?
Let’s talk about the kinds of preventative behaviours.
For example, increasing physical activity, eating a healthier diet, social interaction, brain training, might all help to reduce our risk of developing things like Alzheimer’s and dementia. One of the issues here is that actually sustaining these long term changes and making them long term habits is extremely challenging.
There is quite a good evidence base on which behaviour change techniques work but we’re still really in the early stages of understanding how to implement these techniques in a way that engages different groups of people. There is strong evidence which suggests that we should have a tailored approach.
Thank you for reading!
Food for the Brain is a non-for-profit educational and research charity that offers a free Cognitive Function Test and assesses your Dementia Risk Index to be able to advise you on how to dementia-proof your diet and lifestyle.
By completing the Cognitive Function Test you are joining our grassroots research initiative to find out what really works for preventing cognitive decline. We share our ongoing research results with you to help you make brain-friendly choices.
Please support our research by becoming a Friend of Food for the Brain.
Further info