Why “Normal” Vitamin D Levels May Not Be Protective for Women’s Brains
Why “Normal” Vitamin D Levels May Not Be Protective for Women’s Brains

Science now recognises Vitamin D as far more than a bone-health nutrient. Over the past decade, it has become increasingly clear that vitamin D acts as a hormone regulator, playing an important role in brain health, immune regulation and inflammation, especially when considering optimal vitamin D levels for women’s brains.
What is discussed far less, is a more nuanced question…
If vitamin D matters for the brain, how much is actually enough and do vitamin D levels differ for women?
A growing body of research links lower vitamin D status with a higher risk of cognitive decline and Alzheimer’s disease. Yet most guidance still relies on population-based “normal” ranges that were never designed to protect the brain. This raises an important prevention question for women, who already carry a higher lifetime risk of Alzheimer’s disease.
What the research shows
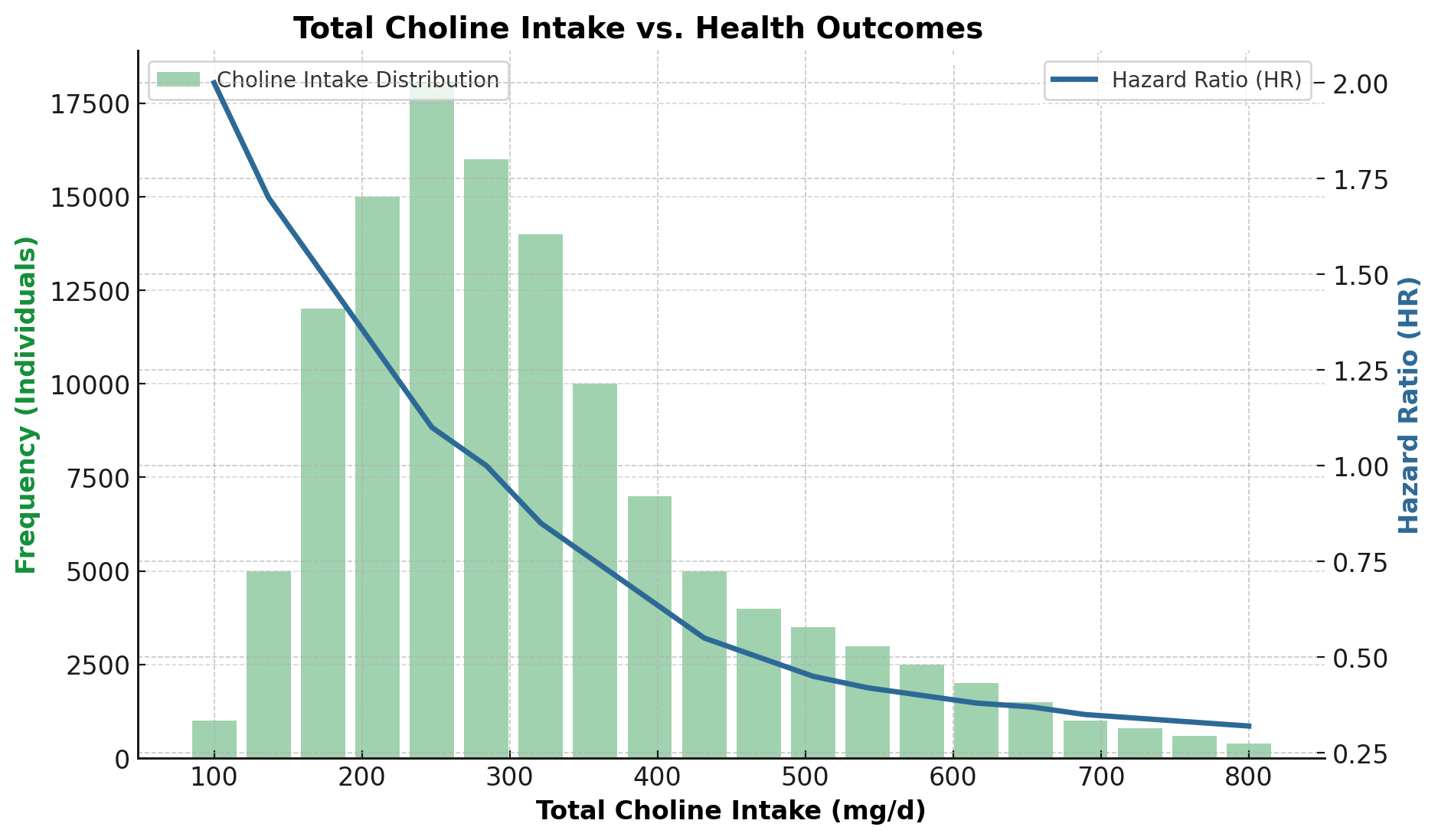
A systematic review and meta-analysis in 2025 by Li et al. examined the relationship between circulating vitamin D levels and Alzheimer’s disease risk across multiple observational studies (1).
The findings were consistent:
• Lower vitamin D levels link to a higher risk of Alzheimer’s disease.
• Risk increased progressively as vitamin D levels declined
• Researchers observed this association across different populations and study designs.
Crucially, the authors did not claim that vitamin D deficiency causes Alzheimer’s disease. Instead, vitamin D status appears to track biological vulnerability in the brain and reflects processes such as neuroinflammation, immune dysregulation and vascular dysfunction, all recognised contributors to cognitive decline.
This distinction matters for prevention.
Why these findings matter particularly for women
Women account for around two thirds of Alzheimer’s diagnoses worldwide. Longevity alone cannot explain this difference.
Across midlife and later life, women experience biological changes that alter how the brain responds to metabolic, inflammatory and hormonal stress. The menopausal transition is a key inflection point. Declining oestrogen and progesterone influence immune signalling, cerebral blood flow and brain energy metabolism, all of which intersect with established dementia risk pathways (3). This helps explain why midlife can be a turning point for brain health in women, even when blood test results appear “normal”.
Vitamin D functions as a hormone-like regulator, with receptors widely distributed throughout the brain and immune system. Its actions include modulation of inflammatory responses, immune balance and neuronal protection. Hormonal changes appear to influence how effectively vitamin D signalling is utilised at a tissue level. This is supported by experimental and clinical research showing interactions between oestrogen, vitamin D receptors and immune signalling, although this is not always directly measured in large population studies. In practical terms, this means that a vitamin D level considered “normal” for the general population may not confer the same degree of neuroprotection in the ageing female brain.
This does not mean vitamin D requirements are definitively higher for every single woman, or that everyone should take high-dose supplementation. Excessive intakes via supplementation over time can be harmful, which is why context, testing and appropriate dosing matter.
Prevention works best when it’s personal, based on what’s happening in your own brain and body, not just what’s considered “normal.”
The problem with “normal” ranges for vitamin D for women’s brains
Researchers established vitamin D reference ranges primarily to prevent overt deficiency-related disease, particularly rickets and osteomalacia. They did not design these ranges to define optimal levels for long-term brain resilience.
Population reference ranges do not account for factors that strongly influence dementia risk, including:
- Chronic low-grade inflammation
- Insulin resistance and blood sugar dysregulation
- Oxidative stress (see our explainer video here)
- Hormonal transitions across midlife
- Genetic variation in vitamin D metabolism and receptor activity
As a result, vitamin D levels that fall within the laboratory “normal” range may still exist within a biological environment that favours cumulative brain damage over time. This limitation is not unique to vitamin D. It reflects a broader problem with single-nutrient or single-cause thinking in Alzheimer’s prevention.
Vitamin D does not act alone in protecting women’s brains
Vitamin D is not an isolated lever in brain health. Low vitamin D status frequently clusters with other modifiable biological risk factors, including:
• Low omega-3 fatty acid status
• Raised homocysteine, reflecting impaired B vitamin-dependent methylation, a process essential for maintaining brain cells
• Poor blood sugar control
• Reduced antioxidant capacity, including glutathione availability
Each of these factors independently links to cognitive decline. More importantly, they interact within the brain.
Alzheimer’s disease does not arise from a single deficiency, a single gene or one pathological protein.
It reflects the cumulative impact of multiple biological systems drifting out of balance over years or decades. This is why interventions that target a single marker so often produce disappointing results.
Prevention requires a broader, systems-based view.
Once you see vitamin D in this broader context, it becomes clear why testing a single marker in isolation can only ever give partial answers.
From nutrients to prevention systems
Testing vitamin D alone can identify a deficiency, but it cannot tell you whether the brain’s key protective systems are functioning together.
A prevention-led approach asks different questions:
- How well is inflammation being regulated?
- Are brain cell membranes supported by sufficient essential fats?
- Is methylation, the nutrient-dependent process that supports DNA repair, neurotransmitter balance and brain structure, functioning effectively?
- To what extent is blood sugar quietly damaging brain neurons over time?
These are not abstract concepts.
They are measurable, modifiable drivers of dementia risk that we assess through our at-home DRIfT blood test.
A smarter way to assess brain health
Many people reading this will have been told their vitamin D is “fine”. They may spend time outdoors, eat well, and still feel tired, foggy or not quite themselves. The problem is not that vitamin D doesn’t matter. It’s that a single number rarely tells the full story.
This systems-based understanding underpins our work at Food for the Brain. It is the heart of prevention.
Prevention is not about chasing one “perfect” nutrient level or one lifestyle change.
It is about understanding how your body works as one connected system and acting early enough to change the trajectory.
If you want to begin supporting and upgrading your brain today:
Complete the Cognitive Function Test today if you haven’t done so yet.
It is free to everyone, validated and provides personalised insights into your current brain health.
Order your at-home DRIfT blood test to assess the key biological drivers of cognitive decline, including vitamin D, omega-3, homocysteine, blood sugar control and antioxidant status. Together, these results give you the information you need to move from awareness to meaningful prevention.
References:
- Li X, et al. The relationship between vitamin D levels and Alzheimer’s disease: a systematic review and meta-analysis.
- Alzheimer’s Association. 2023 Alzheimer’s Disease Facts and Figures.
- Lancet Commission on Dementia Prevention. Livingston G, et al. Dementia prevention, intervention, and care: 2020 report.