Alzheimer’s is the most common cause of dementia, affecting an estimated 850,000 people in the UK. It is characterised by the build-up of two proteins in the brain, called amyloid and phosphotau, which build up and damage brain cells. Amyloid forms plaques outside of brain cells, whereas phosphotau forms tangles inside brain cells. These plaques and tangles lead to the death of nerve cells, which causes areas of the brain to atrophy. Homocysteine, a toxic amino acid, also accumulates and promotes neuronal cell death. When homocysteine levels rise so too do anyloid and p’tau. The hippocampus, which is involved in memory formations, is the first part to be affected. This can cause individuals to have difficulty remembering recent events and retaining new information and experience. However, long term memories are less affected as these are more reliant on other regions of the brain, known to deteriorate later in the progression of Alzheimer’s disease.
Causes
Alzheimer’s disease is very rarely caused by mutations in certain genes and these lead to the early-onset form of the disease. However, these mutations account for less than 1% of all cases. The causes of the common form of Alzheimer’s disease are multifactorial and there have been many attempts to identify the final common path that leads to cognitive impairment and brain atrophy. Clearly, if the nerve cells die then they can no longer support the formation of memories so ultimately we need to find the causes of nerve cell death.
In the 1970s it was found that the acetylcholine-containing neurons in the brain had degenerated in Alzheimer’s disease, which led to the cholinergic hypothesis that cognitive impairment was due to loss of acetylcholine. This led to the introduction of the cholinesterase inhibitors, as the first treatment of cognitive decline in the disease, and these drugs are still the main treatment in use today. However, the cholinesterase inhibitors only provide temporary benefits and do not slow down the disease process.
The amyloid hypothesis proposes that formation and deposition of amyloid-beta as insoluble plaques outside the nerve cells is the main cause and primary driver of Alzheimer’s disease. Most of the drugs being developed and tested today are based upon this hypothesis but they have largely failed to alleviate the symptoms of the disease. In 2021, the US FDA controversially licensed an antibody to amyloid-beta for provisional use. However, the amyloid theory has been subjected to much criticism and its weakness is that, despite some drugs lowering amyloid, none have provduced any clinical benefit. Also, it downplays the significance of tau phosphorylation in Alzheimer’s pathogenesis. Many scientists in the field consider that the main cause of nerve cell death is the formation of neurofibrillary tangles composed of phosphotau which goes up when homocysteine is elevated. Whether phosphotau is the cause or consequence of the disease process is yet to be determined.
Risk Factors
Whatever the final pathway is, the good news is that there are many identified risk factors, which feed into the final path. Some of these risk factors may be modifiable, and could therefore form the basis of approaches which may help to prevent Alzheimer’s disease. Several genetic risk factors have been identified, of which the most important is the epsilon allele of the gene for apolipoprotein E; 15% of people born with the ApoE4 genevariation could have an increased risk of 4%. Overall, the genetic risk factors do not determine that a person will develop the disease and are less important than the non-genetic risk factors.
It can be concluded that the development of Alzheimer’s disease is due to multiple reasons, encompassing genetic, dietary and lifestyle factors. Less than 1% of Alzheimer’s is caused by genes. Thus, nutritional and lifestyle considerations are very relevant when considering not only the pathogenesis of Alzheimer’s disease, but also exploring preventative measures. A good example of the influence of a poor diet is a study from Stockholm, where elderly people were followed for 7 years and their cognitive test scores were found to be related to their dietary habits, with a faster decline in those who adopted a typical ‘Western diet’ (Figure 1).
Figure 1. Relation of usual diet to rate of cognitive decline in elderly (Shakersain, 2016)
Other dietary habits that have been associated with a decreased risk of cognitive impairment in ageing are the regular consumption of foods containing substances with anti-oxidant properties (like flavonoids) such as green vegetables, berries, tomatoes, and fruits. These foods are also a good source of folate, which lowers homocysteine and may be protective against the development of Alzheimer’s disease.
Development and Progression
Alzheimer’s disease typically progresses slowly in three general stages: mild, moderate, severe. On average, a patient diagnosed with Alzheimer’s disease can be expected to live, on average, around four to eight years after diagnosis. However, individuals can begin to develop dementia years before they are actually diagnosed.
There are seven different stages of dementia:
One – no impairment
Two – very mild cognitive decline
Three – mild cognitive decline
Four – moderate cognitive decline
Five – moderately severe cognitive decline
Six – severe cognitive decline
Seven – very severe cognitive decline
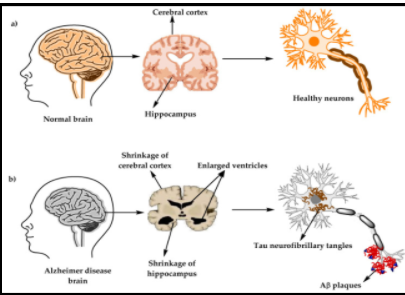
Figure 2. Some differences between Healthy Brain and Brain Affected by Alzheimer’s Disease
Treatment or prevention?
The brain in a person with Alzheimer’s disease has lost as much as 30% of its tissue, mostly nerve cells. Even if it was possible to replace the lost nerve cells, it would not be possible to replace the memories stored in the nerve cell circuits. So claims that a ‘cure’ for Alzheimer’s disease can be developed are wrong. But what we can do is to find ways to prevent or slow down the disease by modifying the non-genetic risk factors.
Prevention must start before onset or at early stages of the disease, such as Mild Cognitive Impairment or mild Alzheimer’s disease, since there is little chance that the disease can be modified when the symptoms are severe enough to give a diagnosis of moderate to severe Alzheimer’s disease. The approach should be based upon the risk factors shown in Figure 1, with the aim of modifying as many of these as is possible.
Approach
It is important for individuals at risk of Alzheimer’s disease to ensure that their diet is balanced with a variety of foods, for example vegetables, fruits, wholegrains, fish and lean meat. Intake of refined sugars, salt and processed foods should be greatly reduced. Dietary patterns are important, as shown by the protective effect of eating a Mediterranean diet which, as well as flavonoids and folate, also contains omega-3 fatty acids. Vegetarians and vegans must ensure that they take supplements of vitamin B12 as this protects the brain from shrinking. Regular physical activity is also important and even a walk of 30 minutes each day is beneficial. Social and cognitive activity are also valuable.
One particular risk factor, raised plasma homocysteine, can be treated by taking supplements of the B vitamins folate, B12 and B6. Moderately elevated plasma total homocysteine is a strong modifiable risk factor for vascular dementia and Alzheimer’s disease. Prospectively, elevated homocysteine is associated with cognitive decline, white matter damage, brain atrophy, neurofibrillary tangles, and dementia. Most homocysteine-lowering trials with folate and vitamins B6 and/or B12 tested as protective agents against cognitive decline were poorly designed by including subjects unlikely to benefit during the trial period. In contrast, trials in high-risk subjects, which have taken into account the baseline B vitamin status, show a slowing of cognitive decline and of atrophy in critical brain regions, results that are consistent with modification of the Alzheimer’s disease process. Homocysteine may interact with both risk factors and protective factors, thereby identifying people at risk but also providing potential strategies for early intervention.
The VITACOG trial in Oxford found that for people with Mild Cognitive Impairment who had a good starting level of omega-3 fatty acids, this treatment markedly slowed the rate of atrophy of the brain and also slowed cognitive decline. The treatment appeared to modify the disease process, reducing the rate of brain shrinkage in those with suffcient omega-3 status by 73% compared to placebo. Trials are needed to see if a combination of B vitamins and omega-3 fatty acids will slow or prevent conversion from Mild Cognitive Impairment to dementia. In the meantime, these nutrients are safe and there is no reason why people with raised homocysteine should not take them after consulting a physician.
Summary
Alzheimer’s disease is a complex disease, characterised by the presence of raised homocysteine, beta amyloid plaques and phosphotau tangles and by the large-scale loss of brain tissue. Many modifiable risk factors have been proposed and there is every hope that interventions to modify these will be able to prevent a significant proportion of cases in the future. From the point of view of nutrition, we can already identify several factors that appear to be preventative but further research is needed to confirm and extend these findings.
Written by: Amy Jade Surman, BSc. Volunteer for Food for the Brain Foundation
Reviewed by: Professor David Smith & Patrick Holford
Republished: August 2022
Next Due for Review: Sept 2023
References
Age UK. Nutritional supplements and cognitive ageing. Available at: https://www.ageuk.org.uk/northern-ireland/information-advice/health-wellbeing/mind-body/staying-sharp/looking-after-your-thinking-skills/nutritional-supplements-and-cognitive-ageing/ [Accessed Sept 6, 2021]
Alzheimer’s Association. Stages of Alzheimer’s. https://www.alz.org/alzheimers-dementia/stages [Accessed Aug 24, 2021]
Alzheimer’s Research UK. Dementia Information. Available at: https://www.alzheimersresearchuk.org/dementia-information/types-of-dementia/alzheimers-disease/?gclid=CjwKCAjw3_KIBhA2EiwAaAAlincCnHQAnZD_yBIte4-haqQNf10kE-hSbF3vjlUzKslKVg0bbHdFFBoCEYUQAvD_BwE [Accessed Aug 24, 2021]
Alzheimer’s Society. Alzheimer’s Disease. Available at: https://www.alzheimers.org.uk/about-dementia/types-dementia/alzheimers-disease [Accessed Aug 24, 2021]
An, Y, et al. Dietary intakes and biomarker patterns of folate, vitamin B6, and vitamin B12 can be associated with cognitive impairment by hypermethylation of redox-related genes NUDT15 and TXNRD1. Clin Epigenetics. 2019; 11: 139 Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6787977/ [Accessed Sept 6, 2021]
Armstrong, R. Risk factors for Alzheimer’s disease. Folia Neuropathol. 2019;57(2):87-105. Available at: https://pubmed.ncbi.nlm.nih.gov/31556570/. [Accessed Sept 6, 2021]
Bekris LM, Yu CE, Bird TD, Tsuang DW. Genetics of Alzheimer disease. J Geriatr Psychiatry Neurol. 2010 Dec;23(4):213-27. doi: 10.1177/0891988710383571. PMID: 21045163; PMCID: PMC3044597.
Breijyeh, Z, Karama, R. Comprehensive Review on Alzheimer’s Disease: Causes and Treatment. Molecules. 2020 Dec 8;25(24):5789. Available at: https://pubmed.ncbi.nlm.nih.gov/33302541/ [Accessed Sept 6, 2021]
Cherian, L. Mediterranean-Dash Intervention for Neurodegenerative Delay (MIND) Diet Slows Cognitive Decline After Stroke. J Prev Alzheimers Dis. 2019;6(4):267-273. Available at: https://pubmed.ncbi.nlm.nih.gov/31686099/ [Accessed Sept 6, 2021]
Counts, S, Laihri, D. Overview of Immunotherapy in Alzheimer’s Disease (AD) and Mechanisms of IVIG Neuroprotection in Preclinical Models of AD. Current Alzheimer’s Review. 2014; 11(7): 623–625. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5870835/ [Accessed Sept 6, 2021]
NHS Inform. Alzheimer’s Disease. Available at: https://www.nhsinform.scot/illnesses-and-conditions/brain-nerves-and-spinal-cord/alzheimers-disease [Accessed Aug 20, 2021]
Kametani,F, Hasegawa, M. Reconsideration of Amyloid Hypothesis and Tau Hypothesis in Alzheimer’s Disease. Front Neurosci. 2018 (12). Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5797629/ [Accessed Sept 6, 2021]
Kaur, D et al., Multifaceted Alzheimer’s Disease: Building a Roadmap for Advancement of Novel Therapies. Neurochem Res. 2021. Available at: https://pubmed.ncbi.nlm.nih.gov/34357520/ [Accessed Sept 6, 2021]
Liu, X, Yang, L, Zhao, L. 2020. Stem cell therapy for Alzheimer’s disease. World J Stem Cells. 2020. 12(8):787-802. Available at: https://pubmed.ncbi.nlm.nih.gov/32952859/ [Accessed Sept 6, 2021]
Nagpal, R, et al. Modified Mediterranean-ketogenic diet modulates gut microbiome and short-chain fatty acids in association with Alzheimer’s disease markers in subjects with mild cognitive impairment. EBioMedicine. 2019 Sep; 47: 529–542. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6796564/ [Accessed Sept 6, 2021]
Norwitz,N.G.;Saif,N.; Ariza, I.E.; Isaacson, R.S. Precision Nutrition for Alzheimer’s Prevention in ApoE4 Carriers. Nutrients 2021, 13, 1362. https://doi.org/10.3390/ nu13041362
Oulhaj, A, et al. Omega-3 Fatty Acid Status Enhances the Prevention of Cognitive Decline by B Vitamins in Mild Cognitive Impairment. J Alzheimers Dis. 2016;50(2):547-57. Available at: https://pubmed.ncbi.nlm.nih.gov/26757190/ [Accessed Sept 6, 2021]
Senior Link. Helpful Daily Activities for Dementia Patients: 50 Expert Tips and Suggestions to Keep Your Loved One Engaged. Available at: https://www.seniorlink.com/blog/helpful-daily-activities-for-dementia-patients-50-expert-tips-and-suggestions-to-keep-your-loved-one-engaged [Accessed Aug 20, 2021]
Shakersain, B, et al. Prudent diet may attenuate the adverse effects of Western diet on cognitive decline. Alzheimers Dement. 2016. 12(2):100-109. Available at: https://pubmed.ncbi.nlm.nih.gov/26342761/ [Accessed Sept 7, 2021]
Smith AD, Refsum H. Homocysteine, B Vitamins, and Cognitive Impairment. Annu Rev Nutr. 2016 Jul 17;36:211-39. doi: 10.1146/annurev-nutr-071715-050947. PMID: 27431367. [Accessed Aug 5, 2022]
Walsh, S. Aducanumab for Alzheimer’s disease? BMJ. 2021; 374. Available at: https://www.bmj.com/content/374/bmj.n1682 [Accessed Sept 6, 2021]
