The Origin of Alzheimer’s Disease
By Patrick Holford
Why Dementia rates are higher in the west
In some countries, for example India and China, that proportion appears to be less than half that occurring in Britain. When people in one country suffer much more from a disease than people of a similar age in another country, this is a sure sign that the difference has something to do with diet, lifestyle or other environmental factors – or genetic variance. We can rule out genetic differences as the major factor, particularly because Chinese and Indian people who emigrate to Britain soon acquire a similar risk for developing dementia. In any event only one in a hundred cases of Alzheimer’s is caused by genes.2
How many people get diagnosed with dementia?
A decline in memory and concentration is not the same thing as a diagnosis of dementia or probable Alzheimer’s, although it does mean your chances of developing these conditions are higher. Every year roughly 10 million people are diagnosed with dementia – that is one person every 3 seconds3 . Currently, around 900,000 people in the UK have dementia.4 By 2050 this will be over 1.53 million. Globally over 50 million have dementia. By 2050 this is expected to increase to 152 million.5
Two in three people diagnosed with dementia will end up diagnosed with probable Alzheimer’s, while 17 per cent will be given a diagnosis of vascular dementia, caused by constricted blood flow to the brain due to blocked arteries, and 10% will be given a ‘mixed’ diagnosis, which is usually part Alzheimer’s, part vascular dementia. But the risk factors, and prevention treatments, for Alzheimer’s and vascular dementia are the same. So, combined, well over 80% of all dementia diagnoses should be preventable.
What is dementia?
There are other forms, such as dementia with Lewy bodies, fronto-temporal dementia and dementia caused by a stroke, a bleed in the brain or a brain tumour. But as Alzheimer’s is the most widespread, let’s look at it in depth.
Dementia – including Alzheimer’s – is an insidious condition. In the early stages, sufferers have increasing symptoms of absentmindedness, low mood and an inability to learn new things. Judgement, and their ability to function intellectually and socially, begin to go awry. The person may repeatedly forget to turn off the iron, or may not recall which medicines they took in the morning. They may start to show mild personality changes, such as a lack of spontaneity or a sense of apathy and a tendency to withdraw from social interactions.
Later on, there will be a loss of logic and memory, disorientation and poor coordination. Speech deteriorates and paranoia may appear. At this point, a diagnosis of probable Alzheimer’s disease may be given. Why ‘probable’? Because Alzheimer’s is properly diagnosed, not simply by symptoms, but by the presence of a specific kind of degeneration in a specific part of the brain – and this is difficult to see without the aid of expensive scans.
How was Alzheimer’s discovered?
The German neuropathologist Alois Alzheimer discovered this characteristic degeneration in the brain back in 1906. Using a technique known as silver stain, he examined the brain cells of a woman who died prematurely at 55 with signs of dementia, and found a tangled mess of proteins and clusters of degenerating nerve endings, called neurofibrillary tangles. This condition is associated with a gradual dying-off of neurons and poor communication between neurons. There is also often a build-up of something called beta-amyloid plaque, a protein-like substance that shouldn’t be there.
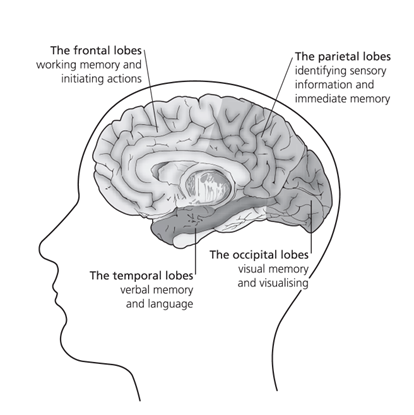
Since that time, research into Alzheimer’s has continued apace. Largely thanks to the pioneering work of Professor David Smith and colleagues in the University of Oxford’s pharmacology department, we now know that Alzheimer’s is a specific disease process, not just a random, gradual decline in brain cells, and that it originates in a particular brain region. Their Optima (Oxford Project to Investigate Memory and Ageing) study has been running since 1988 and has proved, among other things, that the damage leading to Alzheimer’s begins in a central part of the brain known as the medial temporal lobe.6-7
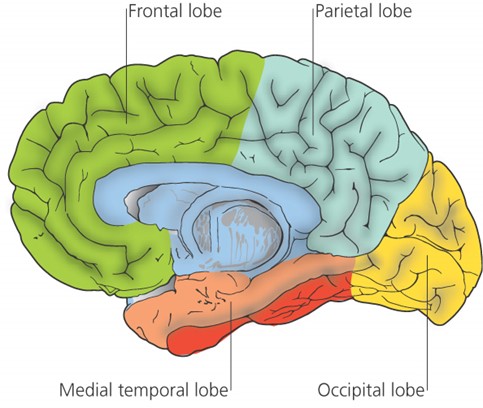
Pinpointing the problem area
The medial temporal lobe is vital for both mood and memory. Even though this lobe accounts for only 2 per cent of the brain’s total area, it is essential for the processing of everything we sense, feel or think.
Precisely because it’s in the middle of the head, it’s a difficult region to scan. This is also where there are more neurofibrillary tangles and beta-amyloid plaques – the hallmarks of Alzheimer’s. These indicate damage and chaos to the normal network of neurons and their connections.
Since information is passed from and to the medial temporal lobe from other parts of the brain, as this area becomes more damaged, fewer signals are sent to other parts of the brain. These then also start to decline, becoming more and more disconnected, with ever-decreasing blood flow. The beginning of damage is estimated to occur as early as 40 years before a person is diagnosed with dementia. That is why it is important to start your prevention plan young.
So far we’ve talked about the spread of damage seen in Alzheimer’s, starting with the medial temporal lobe, and radiating out to other areas of the brain, which are in effect starved of signals, much as a muscle atrophies through lack of use. Other indicators of Alzheimer’s are neurofibrillary tangles (p’tau), the lack of blood flow in the brain, and the presence of beta-amyloid plaques. There is also the presence of high levels of homocysteine in the blood.
Exactly which of these factors ‘causes’ Alzheimer’s, or kickstarts the process of damage, is the subject of much debate and ongoing research.
Clues to curbing the epidemic
At the other end of the spectrum, scientists have been looking for ways to prevent Alzheimer’s disease, and are conducting more and more studies revealing the specific dietary and lifestyle factors that greatly increase or decrease risk. Around half of the risk can be prevented.8For example, having a high intake of omega-3 fats and B vitamins appears to reduce risk, while consuming a lot of sugar increases the risk. The National Institutes of Health attributes 22% of Alzheimer’s to high homocysteine and 22% to low omega-3/seafood consumption.9 What’s more people with pre-dementia with good omega-3 status, given extra B vitamins have a 73% less brain shrinkage than those on placebo.10
Somewhere in the middle, scientists are discovering how changes in diet could cause changes in the brain. An example of this is the discovery of an enzyme that both regulates insulin – the key hormone for keeping your blood sugar in balance – and beta-amyloid. There are, however, many other ways, and growing evidence, that sugar and high carb diet driven by eating junk food damages the brain.

The most exciting discovery is the role of B vitamins and how too little can lead to increases in homocysteine in the blood. Since neither beta-amyloid nor those neurofibrillary tangles can be measured before its too late, the discovery that levels of a simple chemical in your blood could be the best predictor of all is the most welcome news – and it should, in our opinion, have revolutionised the early diagnosis and preventative treatment of those most likely to develop Alzheimer’s. There is good evidence that homocysteine, a measure of faulty methylation, is a primary driver of Alzheimer’s for a number of reasons:

- Giving people with raised homocysteine and pre-dementia (mild cognitive impairment or MCI) extra homocysteine lowering B vitamins has been shown to reduce the rate of shrinkage of the medial temporal regions by nine fold.
- Amyloid blocking drugs have little to marginal effects on the actual disease. A meta-analysis of these drugs that did effectively lower amyloid found virtual no significant cognitive benefit from doing so.11 Measures of Clinical Dementia Ratings show that both homocysteine-lowering B vitamins and Omega-3 fish oil supplements surpass anti-amyloid drugs. (See our newsletter)
- The formation of neurofibrillary tangles, associated with p-tau proteins, could be a consequence of faulty methylation (eg raised homocysteine). When p-tau is high so is homocysteine. There are three known ways whereby raised homocysteine would raise p-tau.
- Homocysteine is found in the regions of brain damage and is capable itself of causing brain damage.
- A raised homocysteine increases the risk of cerebral vascular dysfunction by a remarkable 17 times.11
- Every study that has effectively lowered homocysteine in people at risk, eg with MCI or mild Alzheimer’s, has shown benefit, except in the later stages of the disease which may just be too late.
An International Consensus Statement in 2018 concluded that moderately raised plasma total homocysteine (>11mcmol/L), found in half of those over age 70 12, is a main cause of age-related cognitive decline and dementia.13 Two major meta-analyses of hundreds of studies conclude that raised homocysteine is one of the best evidenced risk factors for AD and accounts for around a fifth of all risk 14, 15.
The key to prevention is to understand the contributing factors and to do something about them as soon as possible. Right now, because the thought of Alzheimer’s is so terrifying, most people avoid even seeing their doctor and are usually diagnosed only in the late stages, usually reported by a relative who has found their partner becoming unmanageable. That’s why it is critical to look for the earliest possible signs of cognitive decline, then there’s time to reverse the trend.
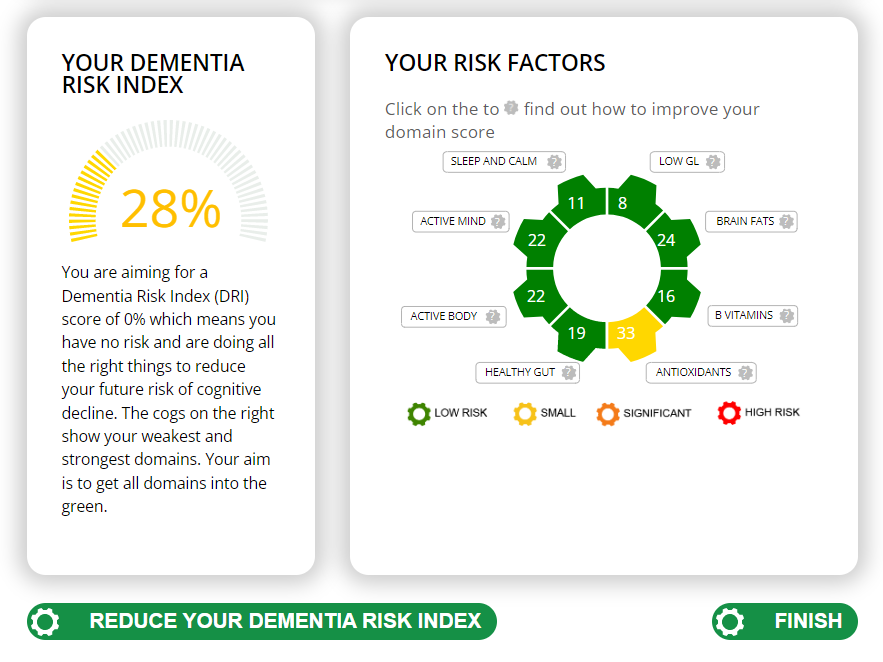
The Cognitive Function Test
The Food for the Brain Foundation offer an excellent free online Cognitive Function test and a simple Dementia Risk Index questionnaire which also works out your risk factors and which simple changes will have the most effect. Do please do this yourself and encourage everyone you know over 50 to do the test as well. Prevention, in this case, is the only likely ‘cure’ for this terrible disease.
For more on Alzheimer’s see our article on Preventing Alzheimer’s Disease
Help support Food for the Brain
Food for the Brain is a non-for-profit educational and research charity that offers a free Cognitive Function Test and assesses your Dementia Risk Index to be able to advise you on how to dementia-proof your diet and lifestyle.
By completing the Cognitive Function Test you are joining our grassroots research initiative to find out what really works for preventing cognitive decline. We share our ongoing research results with you to help you make brain-friendly choices.
Please support our research by becoming a Friend of Food for the Brain.
References
- Rowe J., Kahn R., ‘Human ageing: usual and successful’, Science, 237 (4811): 143-9 (1987).
- Bekris, L et al., ‘Genetics of Alzheimer disease’ Journal of Geriatric Psychiatry and Neurology 2010, 23(4) 213-227).
- Smith A.D., ‘Homocysteine, B vitamins and cognitive deficit in the elderly’, American Journal of Clinical Nutrition, 75:785-6 (2002).
- World Alzheimer Report. (2018). Available online at: https://www.alzint.org/resource/world-alzheimer-report-2018
- Bradley K.M. et al., ‘Cerebral perfusion SPET correlated with Braak pathological stage in Alzheimer’s disease’, Brain, 125:1772-81 (2002); see alsp Jobst K.A. et al., ‘Detection in life of confirmed Alzheimer’s disease using a simple measurement of medial temporal lobe atrophy by computed tomography’, Lancet, 340:1179-83 (1992).
- Jobst K.A. et al., ‘Association of atrophy of the medial temporal lobe with reduced blood flow in the posterior parietotemporal cortex in patients with a clinical and pathological diagnosis of Alzheimer’s disease’, J Neurol Neurosurg Psychiat, 55:190-4 (1992); see also Jobst K.A. et al., ‘Rapidly progressing atrophy of medial temporal lobe in Alzheimer’s disease’, Lancet, 343:829-30 (1994).
- Smith,D., Jaffe,K. ‘Dementia (Including Alzheimer’s Disease)can be Prevented: Statement Supported by International Experts’ Journal of Alzheimer’s Disease 38 (2014) 699–703
- M. Beydoun et al, ‘Epidemiologic studies of modifiable factors associated with cognition and dementia: systematic review and meta-analysis BMC Public Health 2014, 14:64 [http://www.biomedcentral.com/1471-2458/14/643]
- Jernerén F, Elshorbagy AK, Oulhaj A, Smith SM, Refsum H, Smith AD. Brain atrophy in cognitively impaired elderly: the importance of long-chain ω-3 fatty acids and B vitamin status in a randomized controlled trial. American Journal of Clinical Nutrition. 2015;102:215-21.
- Teng Z, Feng J, Liu R, Ji Y, Xu J, Jiang X, Chen H, Dong Y, Meng N, Xiao Y, Xie X, Lv P. Cerebral small vessel disease mediates the association between homocysteine and cognitive function. Front Aging Neurosci. 2022 Jul 15;14:868777. doi: 10.3389/fnagi.2022.868777. PMID: 35912072; PMCID: PMC9335204.
- Smith AD Effect of reductions in amyloid levels on cognitive change in randomized trials: instrumental variable meta-analysis BMJ 2021;372:n156
- Smith AD, Smith SM, de Jager CA, Whitbread P, Johnston C, Agacinski G, et al. Homocysteine-lowering by B vitamins slows the rate of accelerated brain atrophy in mild cognitive impairment. A randomized controlled trial. PLoS ONE. 2010; 5: e12244.
- Smith AD, Refsum H, Bottiglieri T, Fenech M, Hooshmand B, McCaddon A, et al. Homocysteine and dementia: An international consensus statement. J Alzheimers Dis. 2018; 62: 561-70
- Beydoun MA, Beydoun HA, Gamaldo AA, Teel A, Zonderman AB, Wang Y. Epidemiologic studies of modifiable factors associated with cognition and dementia: systematic review and meta-analysis. BMC Public Health. 2014; 14: 643.
- Yu JT, Xu W, Tan CC, Andrieu S, Suckling J, Evangelou E, et al. Evidence-based prevention of Alzheimer’s disease: systematic review and meta-analysis of 243 observational prospective studies and 153 randomised controlled trials. J Neurol Neurosurg Psychiatry. 2020; 91: 1201-9